Leachable substances refer to chemical substances released from medical devices and related liquids, such as monomers, additives, sterilizers, process residues, degradation products, etc. When medical devices are working, leachable substances also produce short-term or long-term safety hazards to human body. Therefore, research on the safety of leachable substances is not only the focus of enterprises in the stage of product design and development, but also the focus of relevant product technology review.
Medical Device Leachables Safety Evaluation Service
- Material selection/process optimization evaluation
- Toxicological risk assessment
- Material equivalent assessment
- New material screening
Basis of Testing Methods
ISO 10993-17, GB/T16886.17
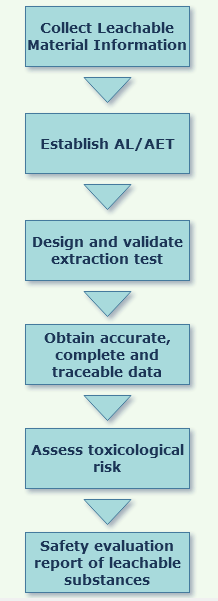
Service Process
Our Advantages
Whole Industry Chain Services|Provide full industry chain technical services, including R&D support, safety evaluation and testing, registration, clinical, quality management system, etc.
Professional Technical Team | A team of experts in the fields of medical device regulations, registration, medicine, toxicology, statistics, biological evaluation, chemical analysis, microorganisms, etc., with comprehensive technical service capabilities
Professional Laboratory|Own several professional laboratories, including chemical analysis lab, materials lab, microorganisms lab, environment lab, efficacy evaluation lab, animal safety evaluation lab, etc.
Rich Industry Experience|Successful cases on safety assessment, registration and clinical services in active, non- active and IVD; Rich experience in implantable degradable products, medical beauty products, oral materials, cavity stents, assisted reproductive products and orthopedic ophthalmology field.
If you have any needs or questions, please contact us at test@cirs-group.com.
